Article in Electric & Hybrid Marine Technology International, April 2019, by Lithium Werks’ CTO, Yazid Saidi.
A density functional theory study underlines the fundamental safety advantages of phosphate technology over layered oxides
Oxide ions, in LiFePO4, as well as in LithiumWerk’ patented LiFe1-yMgyPO4 olivine structure, form a hexagonal close packing (hcp) arrangement. The metal (Fe) ions form zigzag chains of octahedra in alternate basal planes bridged by the tetrahedral phosphate groups (PO4). The lithium atoms occupy octahedral sites in the remaining basal planes. The strong covalent bonding between the oxygen and P5+ to form (PO4)3– units enables greater stabilization of the structure compared with layered oxides such as LiCoO2, where the oxide layers are more weakly bound.
…
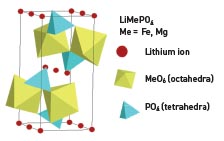
LithiumWerks’ patented chemistry, LiFe1-yMgyPO4, means that by design the amount of lithium extracted is limited. Furthermore it is also generally understood that the transition metal monophosphates are close to refractory in nature, with melting points often far in excess of 1,000°C (1,800°F), adding to their intrinsic safety characteristics under thermal abuse conditions.
Read the article in its entirety here: https://www.ukimediaevents.com/publication/4b21f3e9/84
